
As you can see, there are three blue bubbles of substituents and no lone pairs, meaning the VSEPR notation at this specific carbon is AX 3, meaning it will be trigonal planar. Each blue bubble represents a different substituent group (or atom) coming off of that carbon. We can do the same thing for the carbon second from the right, as shown in the image above. VSEPR predicts this will be a tetrahedral carbon atom as it has the AX 4 configuration of four bonded groups and no lone pairs, as we treat each hydrogen atom as a separate substituent and the everything else residing to the right of the carbon as one substituent. In the example above, we will only examine the carbon furthest to the left. One way you can use VSEPR is to call a group of atoms one substituent. 
Q: Does VSEPR theory work for more complex molecules?Ī: Yes, it can, however, it is important to remember that VSEPR is a tool and has its limits. So….what we need to remember is that for the AX 5 group, you need to replace equatorial atoms with lone pairs AND for the AX 6 group, you need to replace the atoms on the axis with lone pairs, as we have shown above. Thus, we can’t just substitute a lone pair for any old atom. Here, there is a geometric difference between the atoms on the axis (called axial substituents) and the ones around the middle, called the equatorial substituents. It get a little trickier when we get to the 5 and 6 substituent molecules (AX 5 group and AX 6 group, respectively). Same for AX 3E because all of the atoms are geometrically equivalent. So for AX 2E, it is simple to see that we get trigonal pyramidal as the answer because we can replace any of the atoms with a lone pair because they are all geometrically equivalent. In the chart above we have tried to show how this works by just blurring out an atom for a lone pair.įor the 3 and 4 substituent molecules (AX 3 group and AX 4 group, respectively) it is easy to do this because each one of the substituent atoms is the same. Here is one way to remember this chart: Think about each lone pair as just replacing an atom. We know this because of the bond angles associated with each of the four types of shapes. Hence, simple molecules (like the ones we are looking) at will tend to place substituent atoms as far from each other as possible. We also know that electrons repel each other. Let’s not forget, the whole purpose of VSEPR is to minimize interactions between the substituents (atoms and lone pairs) of a molecule. 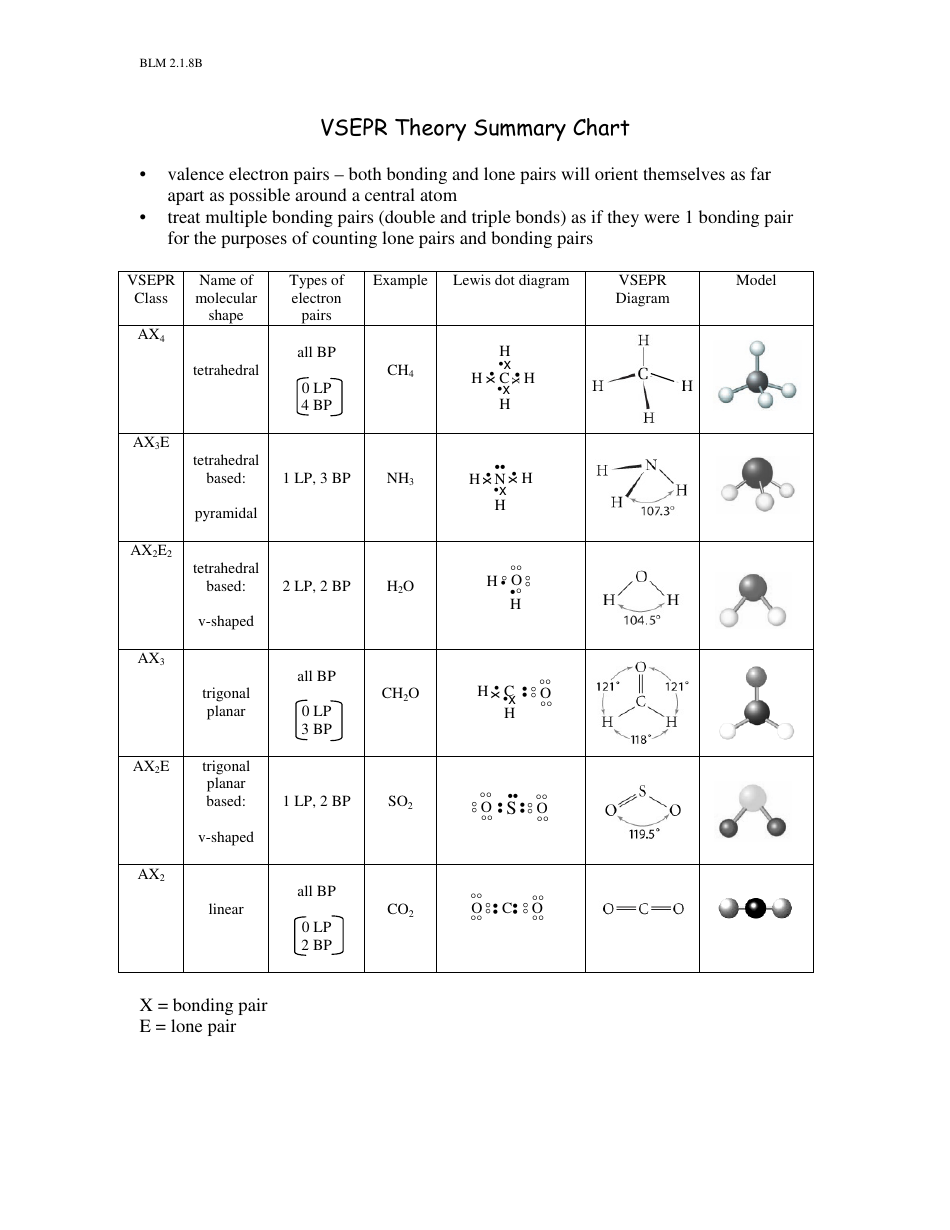
This geometry allows for substituents (atoms and lone pairs) to be as far apart from each other as possible. Each of these will be 120 degrees.Īs you can see from the diagram above, all the bond angles are 120 degrees. There are two Cl-C-O bond angles, and one Cl-C-Cl bond angle. There are two different bond angles in this molecule, but they will both be the same angle. Bond angles help show molecular geometry of COCl 2 As you can see from the chart, AX 3 molecule is trigonal planar. Step 3: Use the VSEPR table to determine the geometry of COCl 2. Step 2: Apply the VSEPR notation to the molecule.įor this one, we can see that it has one central atom, three surrounding atoms, and no lone pair of electrons around the central atom, making it AX 3. Step 1: Determine the Lewis structure of the molecule.įor COCl 2, it is as shown below: For a full-explanation of how to figure out the Lewis structure, please go to Lewis Structure of COCl 2. There is an easy three-step process for determining the geometry of molecules with one central atom. COCl 2 looks like this: How do you find the molecular geometry of COCl 2? Hence, the molecular geometry of COCl 2 only has 120 degree bond angles in the molecule. In this video, we continue exploring VSEPR theory for molecules and ions with five regions or clouds of electrons around the central atom, focusing on examples where the molecular geometry is T-shaped (two of the clouds are lone pairs) or linear (three of the clouds are lone pairs). The molecular shape of COCl 2 is trigonal planar, or AX 3 using Valence Shell Electron Pair Repulsion (VSEPR) theory. Table 1 is the summary of the electron domain geometries and the corresponding molecular geometries.What is the molecular geometry of COCl 2? An example is water ( ), where oxygen is red, and hydrogens are white. If two electron domains are lone pairs, the electron domains geometry is still tetrahedral, but the molecule geometry is bent. An example is ammonia (:NH 3), where nitrogen is blue, and hydrogens are white. If one of the electron domains is a lone pair, the electron domain geometry is still tetrahedral, but the molecule geometry is trigonal pyramidal as with three pereferal atoms at the corners of the triangel and the central atom raised to the top of the pyramid. An example is methane CH 4, where carbon is gray, and hydrogens are white. \)įour-electron groups are farthest apart when they are at the corners of a tetrahedron in a tetrahedral geometry with the central atom at the center of the tetrahedron and the bond angles of 109.5 o around the central atom as.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
